A recent study on Long COVID has uncovered a direct physical relationship between tiny circulating microclots and neutrophil extracellular traps, known as NETs. This connection indicates fundamental biological processes linking the two elements, which could play a key role in the disease when these mechanisms become imbalanced.
These microclots consist of small, atypical aggregates of proteins involved in blood clotting that travel through the bloodstream. The concept gained prominence in 2021 when Professor Resia Pretorius from the Department of Physiological Sciences at Stellenbosch University identified these irregular protein formations in blood samples taken from COVID-19 patients. Their possible role in clotting disorders associated with COVID-19 attracted considerable interest as the global health crisis progressed.
What exactly are neutrophil extracellular traps (NETs)?
Researchers led by Dr. Alain Thierry at the Montpellier Cancer Institute (IRCM) under INSERM in Montpellier were pioneers in emphasizing the significance of NETs in COVID-19 cases. NETs emerge when neutrophils, a type of white blood cell, release their DNA via a mechanism termed NETosis. This results in web-like formations loaded with destructive enzymes that efficiently capture and eliminate invading pathogens.
While NETs serve a vital protective function against infections, their overproduction can lead to adverse effects. An surplus of NETs has been associated with various severe conditions involving inflammation and clotting, such as intense infections, autoimmune disorders, cancer, diabetes, and arthritis. Dr. Thierry points out that continuous excessive NET generation, propelled by recurring cycles of inflammation and clot development, could intensify the progression and severity of illnesses.
Motivated by these insights, the research groups headed by Professor Pretorius and Dr. Thierry collaborated to explore potential interactions between microclots and NETs specifically in individuals suffering from Long COVID.
Primary discoveries from the research
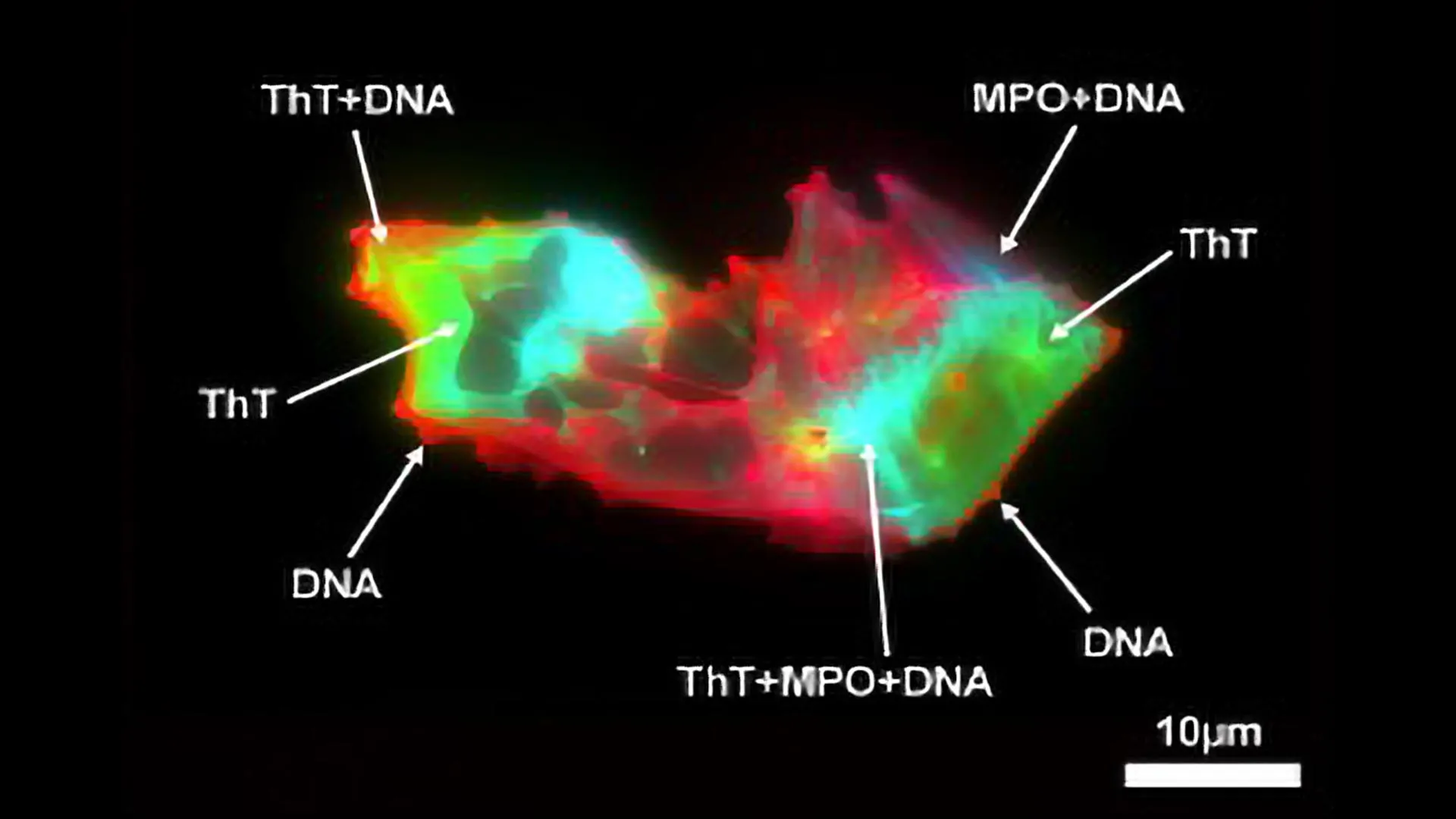
Employing advanced techniques like imaging flow cytometry and fluorescence microscopy, the scientists meticulously evaluated microclots and NETs in the blood plasma of Long COVID patients, contrasting these findings with those from healthy individuals. They further measured NET levels by examining specific protein markers and free-floating DNA in the samples.
The investigation yielded several critical observations:
- Markers indicative of both microclots and NETs were markedly higher in the blood of Long COVID patients.
- Long COVID sufferers exhibited not just a greater quantity of microclots but also microclots of increased size.
- The most striking revelation was the clear structural linkage between microclots and NETs, present across all samples yet dramatically more evident in those with Long COVID.
Dr. Thierry elaborates, “This discovery points to inherent physiological connections between microclots and NETs, interactions that turn harmful when they spiral out of control.”
To deepen their analysis, the team integrated artificial intelligence technologies, such as machine learning algorithms, to scrutinize patterns in the biomarkers. These sophisticated approaches enabled precise separation of Long COVID cases from healthy controls and highlighted the most effective biomarker pairings. Such advancements hold promise for improved diagnostic accuracy and tailored therapeutic interventions in the future.
Professor Pretorius underscores the accumulation of microclots observed in the plasma of Long COVID patients, which appears to be bolstered by heightened NET activity: “This dynamic interplay might make microclots more resistant to natural breakdown processes like fibrinolysis, allowing them to linger in the bloodstream and fuel ongoing issues in the tiny blood vessels,” she details.
Through illuminating the ways in which NETs reinforce microclots, this research sheds crucial light on the underlying biology driving Long COVID symptoms. It also opens doors to potential treatments focused on curbing excessive clotting and inflammatory responses.
Moreover, the study propels efforts to identify novel biomarkers that could enhance the diagnosis and ongoing management of Long COVID. As highlighted by the authors, “Merging state-of-the-art imaging methods with machine learning provides a solid foundation for these findings and enriches the broader conversation around long-term effects of viral infections.”
In Long COVID patients, fresh research has demonstrated a notable structural bond between these persistent microclots in circulation and neutrophil extracellular traps (NETs).
Such a revelation implies deeper physiological ties between microclots and NETs, which, if disrupted, could foster disease progression.
Understanding microclots in detail
Microclots, a term increasingly common in scientific publications, describe anomalous groupings of clotting proteins that float freely within a person’s bloodstream. Professor Resia Pretorius from Stellenbosch University’s Department of Physiological Sciences first brought this phenomenon to light in 2021 upon detecting these unusual structures in blood from COVID-19 sufferers. The finding sparked intense discussion amid the pandemic, given its implications for clotting abnormalities tied to the virus.
Delving deeper into neutrophil extracellular traps (NETs)
The team under Dr. Alain Thierry at the Montpellier Cancer Institute (IRCM), part of INSERM in Montpellier, was at the forefront in recognizing NETs’ pivotal involvement in COVID-19’s disease mechanisms. NETs arise from a unique immune reaction called NETosis, during which neutrophils eject their DNA to weave thread-like networks infused with potent enzymes that swiftly ensnare and destroy harmful microbes.
That said, when NET production goes into overdrive, it can provoke damage, feeding into numerous inflammatory and blood-clotting pathologies like acute infections, autoimmune conditions, malignancies, diabetes, and joint diseases such as arthritis.
Dr. Thierry suggests that unrelenting NET overproduction, sustained by vicious cycles of inflammation and thrombosis, likely amplifies the harshness of the condition.
Through their joint initiative, Professor Pretorius’s and Dr. Thierry’s teams probed the interplay between microclots and NETs within the framework of Long COVID persistence.
Core results of the investigation
By applying imaging flow cytometry alongside fluorescence microscopy, they carried out thorough quantitative and morphological assessments of microclots and NETs in plasma samples from Long COVID patients versus healthy subjects. Quantification of NETs additionally involved scrutiny of associated protein indicators and circulating DNA levels.
- Patient samples displayed substantially elevated biomarkers tied to microclots and NETs alike.
- There was a clear uptick in both the count and dimensions of microclots in affected individuals.
- Crucially, a novel structural connection between microclots and NETs was documented universally, though far more prominently in Long COVID cases.
Dr. Thierry states, “This observation hints at foundational physiological engagements between microclots and NETs that prove pathogenic under dysregulation.”
Additionally, by weaving in AI-driven tools like machine learning for biomarker evaluation, they achieved reliable differentiation between Long COVID patients and controls. The models pinpointed optimal biomarker sets, bolstering diagnostic precision and fostering paths toward individualized care strategies.
Professor Pretorius notes the pronounced buildup of microclots in Long COVID plasma, seemingly anchored by surplus NETs: “Such synergy may shield microclots from degradation via fibrinolysis, extending their circulatory lifespan and exacerbating long-term microvascular dysfunction,” she clarifies.
In delineating NETs’ contribution to microclot durability, the research offers fresh perspectives on Long COVID’s pathological underpinnings. It bolsters pursuits for therapies targeting these intertwined thrombo-inflammatory pathways.
Ultimately, this work lays groundwork for innovative biomarkers aiding diagnosis and treatment oversight: “Integrating cutting-edge imaging with machine learning ensures methodological strength and meaningfully advances discussions on post-viral conditions,” the researchers affirm.