Your genetic makeup is predominantly human, yet it harbors a surprising component from ancient invaders. Approximately 8 percent of your genome originates from viruses that integrated into it long ago. These viral remnants serve as echoes of our evolutionary history, underscoring the longstanding companionship between viruses and humanity since our origins.
In most cases, this 8 percent viral portion of your DNA remains dormant. Researchers refer to it as part of the enigmatic “dark matter” within the human genome.
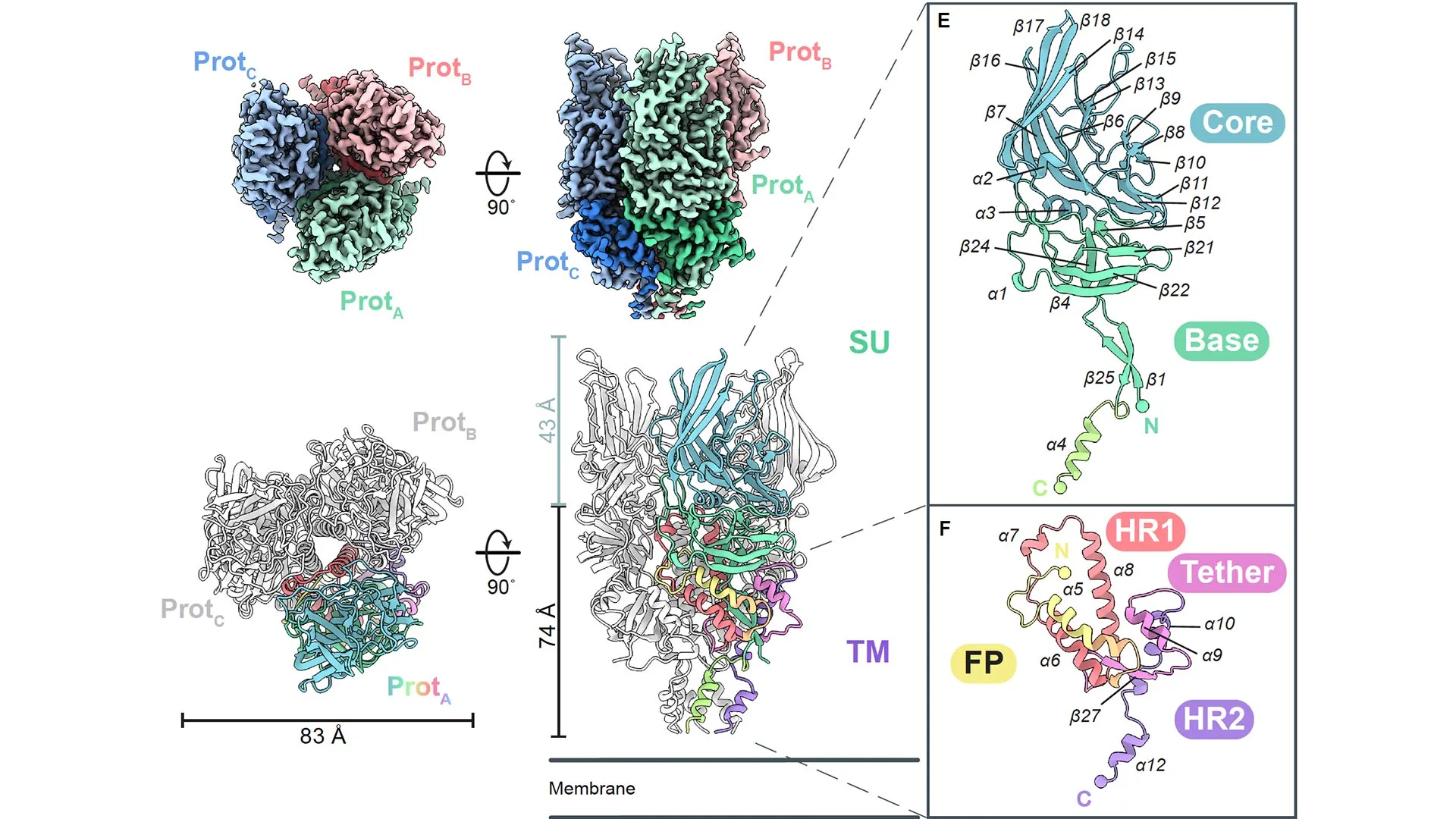
Recently, experts from the La Jolla Institute for Immunology (LJI) unveiled groundbreaking insights into a crucial viral protein. Detailed in a publication from Science Advances, LJI scientists presented the inaugural three-dimensional model of a protein derived from these longstanding human endogenous retroviruses (HERVs).
The research team meticulously charted the surface envelope glycoprotein (Env), which is the primary target for antibodies from the most prevalent HERV variant. This achievement represents a pivotal moment in structural biology. “This marks the very first resolved structure of a human HERV protein—and merely the third retroviral envelope structure determined to date, following those of human immunodeficiency virus (HIV) and simian immunodeficiency virus (SIV),” noted Erica Ollmann Saphire, Ph.D., MBA, who serves as LJI’s President, CEO, and Professor.
Such a revelation paves the way for innovative approaches in disease detection and management. During our distant evolutionary history, HERV-K Env proteins adorned the exterior of HERV-K retroviruses. In contemporary humans, however, these proteins appear on the surfaces of specific tumor cells and in individuals suffering from autoimmune disorders and neurodegenerative conditions, positioning them as prime candidates for advanced diagnostic and therapeutic developments.
“In various pathological conditions, including autoimmune disorders and cancers, these genetic elements reactivate, prompting the production of viral fragments,” explained Saphire. “By comprehending the architecture of HERV-K Env and the antibodies at our disposal, we unlock possibilities for diagnostics and treatments.”
An unexpected structural twist
Prior to this breakthrough, HERV proteins eluded visualization. Their excessive mobility and instability rendered them undetectable even by the most advanced imaging methods. Capturing the structure of HERV-K Env posed particular difficulties, as the LJI researchers had to preserve its fragile “pre-fusion” conformation.
Envelope proteins bristle with latent energy, functioning like coiled springs poised to fuse with a host cell and initiate infection. Consequently, pre-fusion forms are susceptible to abrupt transitions into their post-fusion configurations. “Even a slight provocation can cause them to unravel,” remarked LJI Postdoctoral Fellow Jeremy Shek, who led the effort alongside co-first author LJI Postdoctoral Fellow Chen Sun, Ph.D.
To elucidate the three-dimensional form of HERV-K Env, the scientists implemented targeted amino acid replacements to stabilize the protein without altering its inherent conformation. Saphire’s group has successfully applied this methodology previously to uncover structures of vital proteins from Ebola virus, Lassa virus, and others. Additionally, they identified and analyzed particular antibodies that secured various iterations of the viral proteins.
With the HERV-K Env structures secured, the LJI team employed cryo-electron microscopy—a cutting-edge, high-resolution imaging method—to generate detailed 3D visualizations at critical stages: on the cell surface, during the infection-driving process, and upon binding with antibodies.
While numerous viral envelope glycoproteins form trimers, HERV-K Env stands apart from all previously observed examples, including those from fellow retroviruses. In contrast to the compact, low-profile trimers of HIV and SIV, HERV-K Env exhibits a tall, slender profile. Moreover, its unique folding pattern—the intricate arrangement of strands and helices forming the functional apparatus—deviates significantly from any known retroviral counterpart.
A promising avenue for clinical applications
This LJI investigation establishes new opportunities to harness HERV-K Env beneficially. Insights into its structure and antibody interactions could facilitate the creation of diagnostic instruments and novel treatments.
For instance, a wide array of cancer cells—including those from breast and ovarian malignancies—display HERV-K Env proteins on their surfaces, unlike normal cells. This distinction allows anti-HERV antibodies to differentiate malignant from healthy tissues. As Sun elaborated, researchers might engineer immunotherapies that precisely target HERV-K Env to locate and attack tumor cells. “This approach enables specific targeting of cancerous cells,” Sun affirmed.
Individuals afflicted with autoimmune conditions like lupus or rheumatoid arthritis similarly exhibit HERV-K Env on their cellular surfaces. Certain theories posit that the immune system perceives these anomalous proteins as threats, mimicking a viral assault. In response, B cells produce antibodies directed against HERV-K Env proteins, much like in genuine infections.
“Deciphering antibody recognition of these proteins was daunting due to the absence of structural data and limited availability of effective antibodies,” Saphire observed.
To address this, the LJI researchers developed a comprehensive set of antibodies, illuminating how the immune response engages the protein’s diverse subunits across its conformational states. With this knowledge, scientists can explore interventions to halt detrimental inflammatory responses.
Furthermore, the team evaluated the potential of these antibodies as diagnostic agents for various autoimmune disorders. They applied the antibodies to patient samples from rheumatoid arthritis and lupus cases, successfully identifying HERV-K Env on neutrophils—immune cells implicated in inflammation—after labeling the antibodies with fluorescent markers.
“These antibodies highlighted abnormal HERV expression on neutrophils from patients with rheumatoid arthritis and lupus, but not from healthy individuals,” Saphire reported.
Scientific curiosity surrounding HERVs is surging, with researchers uncovering its involvement in an expanding roster of diseases. “We have the flexibility to investigate any disease of interest via this pathway,” Shek commented.
These endeavors hold promise for enhancing patient care and deepening our grasp of human biology. Ultimately, each of us carries a viral legacy. The moment has arrived to explore and embrace this integral aspect of our identity.